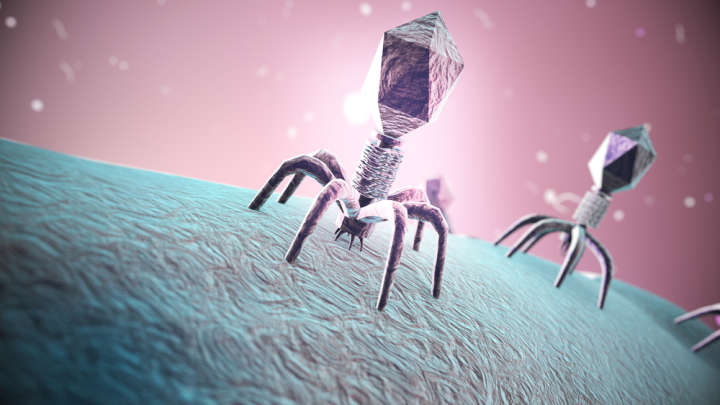
Hundreds Of New Viruses With Enormous Genomes Blur The Boundaries Of Life
Defining life is hard. Plants, animals, and amoebas certainly fit, but what about viruses or prions? For those who put viruses on the non-living side of that division, things just got a bit awkward with the discovery of hundreds of viruses whose genome resembles bacteria.
Bacteriophages are bacteria-infecting viruses. Some have been used in place of antibiotics, but their diversity is so great we’ve barely scratched the surface. Now, a study published in Nature has turned up 351 bacteriophage species with genomes at least four times as large as most members of their class, almost four times the number known when the study began. These viruses have characteristics usually associated with living organisms, blurring the lines between life and non-life.
One genomic giant has 735,000 base-pairs, 15 times as many as a typical phage, beating the previous 596,000 record and more than many bacteria. The paper notes many of these “jumbophages” and “megaphages” are related, suggesting their genome size is an evolutionary stable trait, rather than something that occurred recently.
Large genomes are sometimes full of “junk” DNA, but these viruses are different. These enormous genomes carry parts of the CRISPR system. Now made famous as a method by which humans can edit our own genes with exceptional precision and power, CRISPR originated as a way for bacteria to fight off viral attacks. In the microbial worlds, where genes get shuffled between species, it seems these bacteriophages got hold of CRISPR and turned it to their own ends. Co-first author UC Berkeley graduate student Basem Al-Shayeb thinks the bacteriophages deploy CRISPR to defeat other viruses they battle for control of host bacteria.

These phages inhabit an astonishing variety of locations, from human guts and mouths to soils, hot springs and freshwater lakes, sometimes with closely related jumbophages found in very different environments.
“We are exploring Earth’s microbiomes, and sometimes unexpected things turn up. These viruses of bacteria are a part of biology, of replicating entities, that we know very little about,” Professor Jill Banfield said in a statement. “There definitely seem to be successful strategies of existence that are hybrids between what we think of as traditional viruses and traditional living organisms.”
Combined with this week’s announcement of a virus containing mostly genes we’ve never seen before we’re learning how little we know about the genetics of viruses that don’t infect us.
The team suspects understanding bacteriophages’ CRISPR adaptations will provide pointers for how we can use it ourselves.
Although phages don’t directly infect humans, they can affect our health because they accelerate the transfer of genes between bacteria, including spreading those for antibiotic resistance. Banfield noted that larger genomes are more able to facilitate such gene transfer, so her discoveries may be particularly dangerous, making it important we learn to understand them better.